Triethyl borate came into the world during a period of energetic scientific discovery around organoboron compounds in the early twentieth century. Chemists looked to new elements for applications that would stretch beyond the common materials of the age. The unique characteristics of boron-based esters caught the attention of those studying both organic syntheses and materials that could respond to high temperatures or function as useful solvents. My own first brush with this compound happened in a university lab, running a classic preparation that has long stayed nearly unchanged. Today, the story of triethyl borate represents not only how curiosity leads to solutions, but also how small discoveries ripple into various fields, shaping modern industry and research.
Triethyl borate doesn’t command headlines, but it works quietly behind the scenes. As an organic borate ester, its strengths rest in how it reacts and how it can be transformed. Clear, with a faint odor, the liquid finds its way into labs and manufacturing floors without much fuss. Historically, borates have slipped into everything from fluxes in metallurgy to reagents in chemical reactions. In recent years, the demand for triethyl borate has shifted and grown, as chemists and engineers push for more efficiency, speed, and flexibility in their synthesis work.
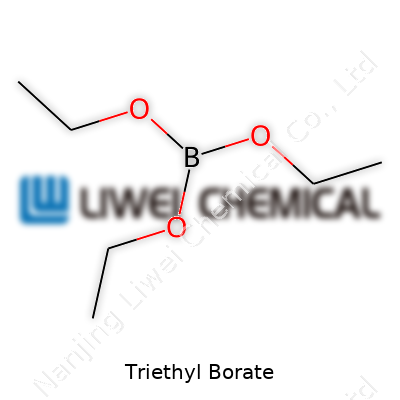
The physical nature of triethyl borate reflects its structure—volatile, flammable, and not very viscous at room temperature. That volatility means it evaporates quickly, which makes it useful for processes needing a temporary solvent or a means to introduce boron into a reaction. The chemical blueprint is simple: three ethoxy groups joined to boron. Its molecular weight keeps it light, and those ethyl groups lend a certain reactivity that synthetic chemists value. Triethyl borate doesn’t dissolve in water, which confines its uses to organic systems but this characteristic also helps guard certain sensitive intermediates from unwanted moisture and hydrolysis.
Talking shop, anyone handling triethyl borate looks for purity markers and a consistency in its physical presentation. It tends to arrive in steel drums or glass vessels, labeled with the proper warnings required for a flammable and mildly toxic substance. Specifications focus on purity, moisture content, and absence of common contaminants, because small deviations in its profile can derail a sensitive synthesis. There are no shortcuts with chemicals like this: handling standards and labeling regulations continue to keep pace with changing safety knowledge and international transport rules. Having worked with a range of suppliers, it's clear that the best ones pay attention not only to the purity, but to documentation and the ease of traceability.
The classic approach relies on a simple exchange—triethyl borate often comes from a reaction between boric acid and ethanol in the presence of an acid catalyst. Water produced by this reaction must be promptly separated, usually through distillation, to shift the equilibrium toward more product. This preparation suits both large industrial production and small-scale laboratory batches, allowing flexibility. The beauty lies in the process’s straightforwardness: a clean starting point for so many more complex reactions.
Chemists reach for triethyl borate when looking to bring boron into the picture. The most famous use shows up in the preparation of organoboranes, essential for organic synthesis and now all but indispensable for Suzuki coupling reactions. Exposure to water or alcohols snaps the molecule apart—hydrolysis yields boric acid and ethanol, both reusable, neither dangerous at this small scale. I’ve watched this hydrolysis countless times; the speed and predictability of the reaction make triethyl borate reliable even in teaching labs. Sometimes, more advanced modifications follow, with substitution or addition, but the value often comes from the initial reactivity of the boron-oxygen bonds.
Depending on where you read or shop, you may see triethyl borate called boric acid triethyl ester or triethoxyborane. The IUPAC names and company trade names change, but the underlying chemistry does not. Recognizing different nomenclature becomes important in procurement and literature searches, especially since regulatory filings and safety data may refer to different synonyms.
Handling triethyl borate means respecting its ability to catch fire and its impact on skin and lungs. Fume hoods are standard, and gloves are necessary. Safety protocols do more than satisfy checklists—they keep accidents from becoming headlines. Storage away from open flames or heat sources remains a rule I never break, after seeing what even a small spill near a hotplate can cause. The compound’s volatility also demands airtight containers, both to prevent atmospheric moisture from creeping in and to stop vapors from accumulating. Training and regular drills matter as much as any technical knowledge, especially as new staff or students enter the workplace.
Triethyl borate lives in many worlds. As a reagent, it makes organoboron compounds for use in pharmaceuticals, agrochemicals, and advanced materials. Flame retardants originate from borate chemistry, and the electronics sector turns to triethyl borate for shells and coatings that need tailored properties. Scientists also use it as a precursor for boron ceramics and specialty polymers, especially where low moisture and fast evaporation help deliver clean products. Laboratory work benefits from its straightforward application in titration and as a solvent with unusual characteristics. In glassmaking, triethyl borate acts as a boron source, changing the thermal and mechanical properties of final products.
The R&D story keeps moving, particularly as sustainability becomes more urgent. Chemists continue searching for greener routes to make and use boron esters, cutting down on waste and energy needs. Novel processes focus on recycling spent materials, using alternative catalysts, or running reactions under milder conditions. In my own conversations with researchers, the constant pressure for higher atom economy and fewer toxic by-products shapes which projects get funding and which fade away. In pharmaceuticals, the quest for new boron-based drugs or imaging agents drives work into previously unexplored reactions.
Research into the toxicity of triethyl borate builds upon the broader understanding of boron chemistry. It isn’t as hazardous as some heavier organometallics, but risks to health do exist. Inhalation of vapors can irritate lungs and mucous membranes, while skin and eye exposure should be avoided. Longer-term effects remain under scrutiny, especially as regulatory agencies tighten exposure limits in response to occupational health studies. Many countries require strict industrial hygiene controls, not just to meet legal mandates but from the lessons of earlier eras when chemical risks went underappreciated. Keeping accurate records of handling incidents and near-misses helps draw a realistic map of where improvements can happen next.
In labs and plants alike, the need for boron compounds continues to grow, especially as demand surges for next-generation materials and greener synthetic approaches. Interest in organic electronics, specialty polymers, and advanced catalysis brings fresh challenges and opportunities. New research aims to reduce both energy use and hazardous by-products, with an eye on stricter environmental regulations. My own hope involves seeing old, reliable compounds like triethyl borate take part in a new wave of sustainable chemistry, helping develop processes that reduce impact while delivering high performance. Investments in purification, automation, and digital tracking support not just efficiency but safety, keeping chemistry labs ready for whatever the next wave of innovation brings.
Triethyl borate doesn’t grab headlines outside chemistry circles, but anyone digging into industrial chemistry, fuel development, or analytical science will run into this clear, sweet-smelling liquid sooner or later. My earliest introduction came during college lab sessions, when I was shocked to learn that some compounds don’t fit easily into just one pigeonhole. Triethyl borate is a quiet example of just that sort of chemical—never as famous as benzene or acetone, but always in the right place at the right time.
Folks who’ve ever watched a fireworks show or even a color-changing fireplace log might have admired triethyl borate’s flair for blue flames. Its most visible use pops up in the world of rocket propellants and flame tests. Drop a bit of triethyl borate into a flame and the result is a striking blue, thanks to the boron content in its structure. In military and aerospace research, scientists use it to produce color markers and signals, or test flame characteristics in controlled settings. Some might shrug at colorful fire, but color provides more than spectacle; blue means boron, and that information can mean safety, a well-run test, or a new discovery about fuel chemistry.
Lab workers depend on the strength and flexibility of certain chemicals to make products, separate mixtures, and carry out sensitive measurements. Triethyl borate excels as a solvent and as a reactant. In organic chemistry, it shows up during the creation of organoboron compounds, which open the door to all sorts of cross-coupling reactions. These reactions form new carbon bonds, driving the creation of pharmaceuticals, agrochemicals, and specialty materials. Cross-coupling might sound like jargon, but it’s basic chemistry at play—linking parts together to build better medicines or stronger plastics.
Triethyl borate’s value grows even more when paired with other boron sources. Anyone making specialized glass or ceramic often seeks boron’s heat resistance and durability. To reach those goals, chemists turn to borate esters like triethyl borate, which slip smoothly into reaction pathways and help introduce boron where it matters. Glass makers and engineers have relied on boron’s properties for generations; triethyl borate is just one more way they get reliable results.
Analytical chemists face a maze of samples every day, and precise measurement matters. Triethyl borate helps in the analysis of alcohols and boron content, serving as a calibration helper in laboratory setups. In fuel labs, it often supports standards for jet fuels—lighter, cleaner burning, and designed for efficiency. The aviation sector, where every drop counts, benefits from this level of precise chemistry.
Triethyl borate’s handling does require respect. It’s flammable, and inhaling its vapors brings health risks. My own reminder came after a long afternoon in the lab, with a whiff that stuck in my nose far longer than I’d hoped. Good ventilation and careful storage prevent a bad day from becoming worse. Most folks outside a lab will never see this chemical, but even a remote risk demands respect, especially where industrial quantities are involved.
Some agencies keep track of triethyl borate because of its flammable nature and potential for harm. The push for greener chemistry and safer industrial practices keeps pressure on researchers and manufacturers to innovate. Long term, the search continues for less hazardous, more sustainable boron sources. Until new options step into the spotlight, triethyl borate will keep plugging away quietly, powering blue flames and laboratory breakthroughs where its unique mix of properties gets the job done.
Understanding chemicals like triethyl borate brings us closer to safer labs, better products, and brighter ideas—not bad for a liquid few folks will ever see outside a lab coat pocket.
People in science classrooms sometimes just want the answers, and for Triethyl Borate, the chemical formula is B(OCH2CH3)3 or more commonly written as C6H15BO3. That’s what you’ll see on bottles, in research papers, and among chemists working in labs. The formula itself packs a story about what Triethyl Borate can do in the lab and in industry.
Back in college chemistry labs, Triethyl Borate showed up now and then—its sharp, almost fruity odor stands out. Among organic compounds, it doesn’t take the spotlight, but it pops up in the right reactions, especially where boron matters. Those hands-on lab moments taught me that learning the formula is only the door to something bigger—understanding what this compound brings to the table in real-world contexts.
Triethyl Borate carries more than just an appetite for reactions; the formula hints at its stability and solubility. Folks working with resins, specialty glasses, and some fire retardants will recognize it. Its boron-oxygen backbone fits into flame-retardant applications, where boron often stops flames from spreading. In synthesis, people sometimes use it as a starting material or as a solvent, especially when precise reactivity is needed.
The chemistry world doesn’t operate on formulas alone. Knowledge about Triethyl Borate gets important when safety steps in. For regular folks, that includes storing the bottle away from open flames, since it’s flammable, thanks to all that ethyl dangling from the boron. Students and researchers know that a chemical’s formula doesn’t just point to what it is—it signals what could go wrong. Rushing into work with any borate, including triethyl, can lead to serious health effects if people ignore proper handling practices.
Google’s E-E-A-T principles matter in chemistry, too. Chemistry knowledge isn’t just about memorizing a string of letters and numbers. Triethyl Borate’s formula stands out because it grounds decisions in the lab or at the job site. Professionals need trustworthy information, and that means drawing lines from established science—chemistry textbooks, peer-reviewed research, practical experience—and sharing that with clarity and confidence. Students pick up formulas but need help making sense of why certain compounds like Triethyl Borate earn their place on the shelf.
This speaks to a broader issue: Basic chemical literacy. Too many people stop at remembering formulas or names, never digging into the practical risks, uses, or environmental impacts. There’s a real need for science education that covers not only what Triethyl Borate does, but how chemicals travel from lab innovation to factory production, or even into waste streams. Teachers and industry mentors have an opportunity to show people how chemical formulas connect with safety, environmental care, and inventive thinking. That’s where chemistry shifts from trivia to essential, trustworthy knowledge—something worth building on for everyone’s safety and progress.
Almost no one thinks twice about all the chemicals used to create products we count on daily. Sometimes a compound like triethyl borate sneaks into labs, manufacturing floors, or even classrooms, and most people never learn its full story. This lack of attention can spell trouble, especially when flammability and toxicity wait in the wings.
Triethyl borate catches fire with surprising ease. It’s got a low flash point—the kind firefighters worry about—meaning just a little spark can set it off. If a workplace stores drums of this liquid near open flames, or even an overheated motor, the risk jumps. In my time working in a busy lab, I saw solvents light up quicker than anyone imagined. People tend to underestimate liquids, thinking only vapors cause real trouble. One misstep with triethyl borate, and you risk not just ignition but an intense burn that spreads fast.
People often store these chemicals alongside other flammable liquids. If a fire erupts, it could feed on several sources at once, overwhelming fire suppression systems. In one facility, a leaky container almost led to a disaster when someone rolled in a metal cart that sparked against a floor grate. Quick thinking and clear safety rules kept it under control that time, but not everyone gets so lucky.
Flammability stands out, but other hazards stick around, too. Breathing in triethyl borate vapors, even for short periods, can irritate the eyes, nose, or throat. It doesn’t take much more exposure for nausea or headache to hit. I’ve watched coworkers shrug off the smell, only to feel queasy later in the day. As with many solvents, skin contact shouldn’t be ignored. Once, a friend splashed his glove and let it slide—redness and skin peeling followed. Cutting corners with chemical safety gear just isn’t worth it.
Folks working with this compound deserve working fume hoods and proper ventilation. Without that, vapors hang in the air, making problems worse. Safety data for triethyl borate isn’t as well known as for some industrial solvents, adding a layer of risk. It takes active training and signage to keep workers sharp and cautious.
Accidents involving flammable chemicals often start with shortcuts. The key to stopping them is culture, not just equipment. In places that stress checking container seals, labeling everything, and giving routine safety talks, I’ve noticed fewer close calls. Personal stories shared in safety meetings make hazards real—no one wants to become the next example.
Government regulations set strict storage and handling standards for flammable liquids, and for good reason. Fire-resistant storage lockers and electronic vapor alarms prove their worth every time they catch a leaking container or overheating drum. Keeping emergency eye wash stations at arm’s reach also shows a commitment to people’s well-being, not just compliance.
Many forget that chemistry delivers both promises and perils. Triethyl borate fits the pattern: its flammability and toxic vapor risks demand practical respect, not fear or indifference. I’ve seen the aftermath of burned lab benches and sick workers. Thorough training, sturdy gear, and careful habits turn a risky chemical into a manageable one, and that’s the sort of outcome everyone deserves. Without vigilance, hazards don’t stay theoretical—they become costly lessons hard-learned.
Triethyl borate sounds like the sort of thing you’d only come across in a lab or an industrial plant. The truth is, lots of folks who work with solvents, specialty glass, or chemical synthesis run into it all the time. This isn’t some inert powder that lets you forget it’s sitting on your shelf. Triethyl borate has a habit of catching fire if you treat it carelessly. That’s something I learned pretty quick experimenting in an old university building with creaky ventilation and a pile of forgotten glass bottles. If someone didn’t label their triethyl borate well, they’d often get a harsh reminder of what a vapors-and-spark combination can do.
Plenty of chemicals demand storage away from heat, but triethyl borate is even fussier than, say, isopropyl alcohol. Its flash point is low. Vapors hang close to the ground and wait for static, a warm bulb, or faulty wiring to bring trouble. It only takes a small lapse—an ungrounded drum, a loose cap, or a door left ajar—to have fumes float into a place they don’t belong.
Watching a container of triethyl borate sweat on a hot summer afternoon made me rethink how to use storage cabinets. The advice to keep it airtight and sealed always makes sense in theory. In practice, a worn gasket lets out enough vapor to make you cough after five minutes in a cramped store room. The headaches or burning eyes are the warning. Listen to them.
I’ve watched well-funded labs fail at safety just as often as small outfits. Pricey explosion-proof fridges don’t help anyone if you let containers get crusted shut or keep incompatible materials close by. Fumes become a bigger issue when storage gets lazy. I learned pretty quickly that storing bottles upright, with leak-proof caps and outside of sunbeams, works better than any protocol printed in a training pamphlet.
Room temperature sounds safe until you remember many buildings heat up unpredictably. Any storage should keep triethyl borate away from spots where temperatures swing or light pours in. Storing it above head height only adds risk since reaching for a heavy glass bottle while balancing on a chair drips danger onto the floor. Best to dedicate a grounded, ventilated, metal cabinet—one used only for flammables. Putting a box of clean-up sand nearby makes sense, not just for rules, but because accidents happen when the regular spill kit runs empty.
Labels seem dull until you inherit a shelf of dusty containers. Sometimes the only thing stopping a mistake is a warning in clear letters. Mark the date you opened a container, since a bottle that’s been around a year or two often gets brittle or forms dangerous peroxides. If triethyl borate sits near acids or oxidizers, even small leaks risk starting a reaction you won't see coming. Strong habits around labeling and sorting make more difference than locking everything behind a heavy door.
Most times, the urge to take shortcuts creeps in near the end of a busy shift. Tucking a container somewhere “just for today” starts the habit of treating risk as routine. All it takes is setting aside the right shelf, keeping it strictly for flammables, and making sure people know why. Folks rarely remember elaborate rules, but everyone understands the memory of a near-miss or a burned finger. Those close calls stick, and they shape how you do things the next time a tricky chemical shows up or the labels start to fade.
Triethyl borate sounds like something that only a chemical engineer or a synthetic chemist ever worries about. But folks working in places where this colorless liquid gets used—like making specialty glass, solvents, or even fuels—know it’s more than another bottle on a shelf. This stuff burns with an almost invisible flame and packs a chemical punch if you treat it casually.
I’ve watched a seasoned coworker get careless with volatile chemicals, and it reminded me how easy it is to let routine dull your sense of risk. Triethyl borate evaporates fast and its vapor can catch fire before you smell anything. You don’t always see flames with it, which means small leaks can turn ugly fast if open flames, sparks, or even hot surfaces show up nearby. I once saw someone in the lab set off a brief, scary flash fire by uncapping a flask too close to a hot plate heating mantle.
Getting it on clothes or skin brings another set of worries. This ester doesn't just dry out your hands like alcohol; it can irritate or burn with enough contact. Breathing in the fumes over time could mess with your airways and knock your plans sideways for days.
Pulling on gloves is the bare minimum. Go for chemical-resistant gloves—nitrile offers solid protection, latex sometimes lets solvents seep through. Safety goggles matter because splash injuries can turn a day in the lab into a ride to the emergency room, especially since boron compounds don’t play nice with eyes. Full-length lab coats and closed shoes round out the basics, but I’ve always preferred a face shield if pouring from larger stock.
Nobody likes working under a clunky fume hood, but for liquids like triethyl borate, that setup isn’t overkill. Just leaving a beaker open on the bench lets vapors drift straight into your breathing space. Decent fume extraction makes inhalation risks much lower. I prefer a sash all the way down, with only arms inside. I remember a day the building ventilation failed and the whole team ended up with headaches before lunch—subtle vapor buildup is a real thing.
Tossing a bottle anywhere in the supply closet makes for a bad surprise later. Triethyl borate likes flammable storage cabinets—away from heat and strong oxidizers. Never store it near acids, since unexpected chemical reactions do not belong in storage rooms. If a spill happens, don’t grab paper towels like it’s water; use absorbents made for flammable solids and make sure the area clears out quickly to avoid any lingering fire risk.
Reading a few hazard labels never replaces hands-on training. Nobody gets the hang of safe handling from a checklist. Practice helps you notice when a seal looks sketchy or if your PPE has pinholes. Clear up confusion fast—years ago, a new coworker tried to mop up a spill with an already damp mop, setting off a small fire. Knowledge isn’t just important, it keeps you from ending up in incident reports.
Every chemical brings its own risks and routines, but triethyl borate asks for focus and respect each time you reach for the bottle. Safety precautions aren’t just about ticking off boxes—they’re about going home without a story to tell about the one time things went wrong.